Exhibiting at the EUMS 2024 show


Presentation at the DFSG 2024



We enjoyed watching Jemma present on behalf of Kettering General Hospital NHS for the SteriPlas treatment of chronic diabetic foot ulcers at the Diabetic Foot Study Group (DFSG). Our Premium Cold Plasma has proven efficacy and anti-biofilm properties essential for the healing of these complex wounds.
Day 2 of the DFSG 2024


Glad to be at the Diabetic Foot Study Group (DFSG) conference catching up with existing customers and to those new who have learnt the benefits of our SteriPlas Premium Cold Plasma. We look forward to the Kettering NHS presentation later today featuring the SteriPlas efficacy for chronic Diabetic Foot Ulcers.
Exhibiting at the DFSG 2024

Cheaper and Faster than Conventional Therapies
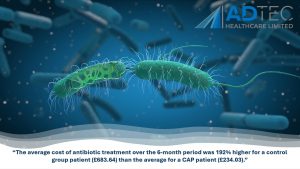
“The average cost of antibiotic treatment over the 6-month period was 192% higher for a control group patient (£683.64) than the average for a CAP patient (£234.03).”
The annual National Health Service (NHS) cost of wound management in the UK for 2017/18 was GB£8.3 billion. Of this, £5.6 billion was spent on managing unhealed wounds (Guest et al., 2020), including diabetic foot ulcers (DFUs) (Everett and Mathioudakis,2018). In 2014-2015 costs for ulceration and amputation in people with diabetes (PWD) was estimated at between £837 million and £962 million with more than 90% of expenditures related to ulceration (Kerr et al 2019).
In the USA, about 38 million Americans have diabetes, and each year a staggering 154,000 Americans will suffer amputations, roughly 80% of which will be the result of complications from diabetes. Their life expectancy following this procedure is five years.
Our mission has always been to revolutionise the global infection market. Bringing an innovative technology which aims to disrupt the conventional infection management market by delivering a more reliable, faster, and effective means of treatment. The SteriPlas has been doing exactly that since its launch many years ago and has expanded to cover the UK, EU and SE Asia with more plans to expand into new territories bringing the benefits to others who have yet to use our medical device.
For more information about our strong clinical efficacy and Health Economics, contact us at info@adtecplasma.com
Adtec joins the Innovation Platform at LSI 2024

We look forward to participating at the Innovation Platform at the @LSI USA 2024. Our CEO, Mary McGovern, will be in the Private Partnering Area between 18-22 March 2024 meeting with exclusive Medtech and Healthtech leaders to bring the rich content of the SteriPlas Premium Cold Plasma to them.
The SteriPlas is renowned for its proven clinical efficacy destroying chronic and complicated biofilm within infected wounds and surgical site infections. Our wide clinical bibliography of 80+ clinical trials and studies ranges from diabetic foot ulcers, LVAD infections, Sternal wound infections and so on.
Delivering a unique physical mode of action during treatment, the SteriPlas dense Cold Plasma ensures the total destruction of all bacterial species protected within biofilm which would otherwise have been resistant to antibiotics. Essentially, we are saving limbs !
We have documented the cost savings benefit due to be published this year across two separate papers focused on diabetic foot ulcers and sternal wound infections.
Our purpose of the LSI Innovation Platform attendance is to seek the potential of investment to help bring our medical device to new territories that have yet to benefit from what we can offer.
Cold Plasma is changing the norm of modern medicine.
Adtec's World Map

Launching an innovative and unknown technology on the medical market has only been successful due to the many years of clinical evidence and trust that we have accrued. From being the first company worldwide to introduce Cold Plasma on wounds in clinical trials to now being the leading manufacturer behind the most effective Cold Plasma medical device, Adtec Healthcare Limited has the strongest experience on how to successfully combat infections stalled by biofilm.
Our global presence of the SteriPlas Premium Cold Plasma is increasing with multiple clinics and distributors endorsing our medical device for chronic infection management of wounds, surgical site infections and medical dermatology.
There are multiple clinics in each country that help to spread the word of the benefit that our medical device brings to non-healing infections. Unfortunately, there is not enough space on this small map to reflect this!
We have also significantly increased our Distributor portfolio into new countries who are rapidly bringing the SteriPlas to their clinics and customers, with plans to expand even further into new territories which we are currently working on.
To learn more about the successes of our dense and rich Cold Plasma, send us a message at info@adtecplasma.com. We are also keen to reach out to new distributors who also wish to bring our Cold Plasma to their territories.
Effective at deep and difficult to treat infection areas
The Premium Cold Plasma from our SteriPlas can kill all bacterial species, regardless of the resistance profile or if they are protected within biofilm. Due to the dense and rich Cold Plasma created, this includes treating difficult to reach areas such as LVAD drivelines, catheters and stoma infections.
Propelled by the gas flow, the Cold Plasma will travel down the gaps around the crevices of the infected driveline until it reaches the site of biofilm which is typically deep underneath the skin fold.
The enhanced physical mode of action of the SteriPlas is delivered quickly and effectively, eradicating bacteria which would otherwise have been resistant to conventional therapies such as antibiotics. These benefits are widely documented including the safety of having no side effects associated to our reliable and consistent Cold Plasma.
The adoption of the SteriPlas has been significantly increasing across the globe for LVAD infection treatments with promising results. To learn more about the SteriPlas, send us an email at info@adtecplasma.com.
The Power of Going Large

The SteriPlas boasts a wide clinical bibliography of clinical trials and publications, documenting the strong clinical efficacy coupled with no side effects reported. This benefits to patients suffering from non-healing wounds infected with biofilm whereby quick 2 minute treatment sessions are able to destroy all bacteria present and heal patients in as quick as 1 week.
To make such a statement as the above requires strong, reputable, and quality-driven results which we are thankful to have. But to collect these results and optimise the technical build of the SteriPlas has taken many years. From our first ever clinical trial on wounds approximately two decades ago to today, the SteriPlas has had its fair share of changes.
Whilst there have been multiple beneficial changes, the overall size of the SteriPlas has remained the same for a crucial reason. The Steriplas is the size it is to produce the optimum plasma for clinical efficacy using microwave argon plasma. Whilst Cold Plasma can still be generated with various technologies, it cannot be done in the same dense and rich manner delivering antibiofilm results. Crucial components to ensure the consistency and reliability may be lost. Most cold plasmas will have antibacterial efficacy against planktonic strains in in-vitro non-competing environments, but chronic wounds are a complex topography of biofilm, exudate, slough, living and necrotic tissue.
Adtec Plasma Technology has developed many Cold Plasma devices since its existence from small pens, portable shoulder strap machines to large medical devices. Our legacy device, the SteriPlas, remains the clinical leader in Cold Plasma medicine for wounds, surgical site infections and medical dermatology conditions.
Seasons Greetings!

Seasons Greetings!
May the new year be even more productive and cheerful than the previous one. Thank you to all our customers, distributors, suppliers and to everyone who has supported us. We would not be where we are today if not for you. Wishing you all the best this season and always.