SteriPlas will be presented at the ISMCS 2023

Congratulations to Mr Thomas Schlöglhofer who has been invited to speak at the International Society for Mechanical Circulatory Support Scientific Congress (ISMCS) on Wednesday 1st November 2023.
His presentation, “Cold Atmospheric Plasma for Driveline Infection Treatment” will focus on the benefits of the SteriPlas for LVAD infections.
Our medical device has proven efficacy for the treatment of deep LVAD infections with the presence of biofilm. There is growing interest to use our SteriPlas Cold Plasma for LVAD infections due to its quick ability to destroy bacteria protected within biofilm deep within driveline cavity. This is regardless of the type of bacteria or its resistance profile. As LVAD infections can be healed in as quick as 1 week, it provides stability of the independent system and a better quality of life for patients with LVADs.
For more information about the SteriPlas, our clinical evidence or a free trial at your hospital contact us at info@adtecplasma
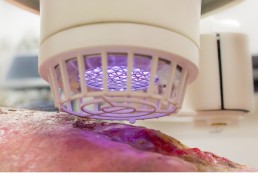
Leading Cold Plasma medicine with the SteriPlas



The SteriPlas produces a repeatable, reliable, and highly efficacious Cold Plasma that has been proven in clinical trials and studies to destroy bacteria protected deep within biofilm.
Regardless of the resistance profile of bacteria, whether they are Gram-negative or Gram-positive, the physical mode of action of the SteriPlas Cold Plasma ensures the destruction of bacteria at the site of infection. This is particularly beneficial for difficult-to-treat or non-healing wounds such as diabetic foot ulcers, sternal wounds and LVAD infections. Where biofilm typically complicates the treatment options for these infections, the SteriPlas has been shown to fully heal patients even if they were not responding to antibiotic treatment to begin with thus saving limbs and lives.
All of this is achieved with the reputable safety of SteriPlas with no side effects reported.
SteriPlas to be presented at the ISDF conference
We’re looking forward to supporting Dr Pierides’ presentation at The International Symposium on the Diabetic Foot (ISDF) conference this Friday as he presents the strong benefits of our SteriPlas Cold Plasma medical device for the management of chronic Diabetic Foot Ulcers. His presentation, “DGH experience of using cold argon plasma in treating diabetic foot ulcers and its role in antibiotic stewardship” discusses the outcomes of 37 patients all of whom received once weekly SteriPlas treatment. The SteriPlas is shown to eliminate the use of antibiotics for chronic DFUs inhibited by multi-resistant bacteria. The unique physical mode of action that the SteriPlas delivers to the infection site ensures a total destruction of all antimicrobial resistant bacteria even if they are protected within biofilm.
For more information about our clinical evidence and medical device, please contact us at info@adtecplasma.com
Day 2 of EWMA
It's day 2 at EWMA 2023 and we're having a great time meeting with new and existing customers. If you want to learn about the most effective Cold Plasma medical device for woundccare with the most clinical trials, strongest efficacy and no side effects, please visit our booth at no. 116 to see a live demonstration of the SteriPlas in action.


How does the Adtec SteriPlas work so effectively?
Our patented cold plasma therapy utilises a physical mode of action approach delivered from an indirect microwave plasma source. Our cold argon plasma is an electrically charged and ionised gas that consists of charged particles, UV light and reactive oxygen and nitrogen species with the latter created as a by-product. They all work collectively to physically rupture the structure of bacteria even if they are protected within biofilm. Regardless of the type of bacteria, its resistance profile or whether they are Gram-positive or Gram-negative our cold plasma works quickly and effectively to destroy them. This has the leading advantage over conventional therapies such as antibiotics that rely on a chemical mode of action approach.
Although antibiotics have been a pivotal part of today’s medicine, they also contribute towards antimicrobial resistance rates particularly because bacteria can develop resistance such as those observed with diabetic foot ulcers that can often lead to amputations as a result.
However, as well as being fast-acting and effective, our cold plasma has shown that a primary or secondary resistance is unlikely to be developed due to its physical mode of action.

Mr Schlöglhofer to present SteriPlas at the ISHLT
Congratulations to Thomas Schlöglhofer from the Medical University of Vienna for his continued success of using our medical device to treat infections in LVAD patients. He will be presenting at The International Society for Heart and Lung Transplantation (ISHLT) conference in April to deliver the great results he has so far achieved.
His presentation, “Cold Atmospheric Plasma Therapy: A Powerful Tool for Treating Driveline Infections in Left Ventricular Assist Device Patients” will be during the “Bugs, Drugs, and Thugs: Fighting Infection in VADs Any Way We Can” Session on Wednesday 19th April between 5:30 PM - 6:30 PM.
We hope you will be there too to see him deliver his promising results.
For more information on how our medical device is fighting against multi resistant bacteria and biofilm in LVAD patients.

Today at The DGTHT Hermedizin 2023
It was a pleasure to see Dr Rotering’s presentation today at the DGTHT Hermedizin 2023 conference. His presentation on the SteriPlas for the treatment of deep sternal wound infections shows just how effective our Cold Plasma medical device is for the treatment of these chronic and deep wounds infected with biofilm.
Not only has the SteriPlas has shown to significantly decrease hospital bed occupancy, but it has shown to reduce mortality rates and prevent the need for expensive treatments by allowing sternal wall stability. The SteriPlas can successfully kill all forms of bacteria, regardless of their resistance profile or if they are protected within biofilm.






SteriPlas heals wounds quickly

Did you catch the leading Serbian media newspaper, Večernje Novosti, last week? Our SteriPlas was featured in the article, “Cold plasma heals wounds quickly” composed by Dr Zorić.
Dr Zoric has been using our cold plasma medical device to treat hard-to-heal and stalled wounds infected with biofilm. These problematic wounds have responded very well with our SteriPlas treatment and patients have been healed that were otherwise stalled by conventional therapies.
If you are within Serbia and interested to learn more about our medical device, contact our medical distributor Borf Health Care on info@borf.biz for more information.
Dr H. Rotering presenting SteriPlas DSWI efficacy at Herzmidizin 2023 conference

Dr Heinrich Rotering will be presenting at the Herzmedizin 2023 conference on 14th February 2023. His presentation, “Tissue saving – Plasma based treatment concept for sternal infection including one year follow-up” will show the benefits of our SteriPlas cold plasma medical device for the treatment of deep sternal wound infections.
The benefit our cold plasma device has for these chronic wound infections is the ability to give a deep penetration treatment allowing plasma coverage over the wide infected area. As our medical device is the only microwave-powered, argon cold plasma device with a wide 12cm2 treatment size and is contact-free, the overall sessions are quickly completed. The mean wound healing for deep sternal wound infections is 16 days with the SteriPlas.
Contact us at info@adtecplasma.com to learn more about the SteriPlas.
Cold Plasma Diabetic Foot Ulcer Clinical Trial Published
Congratulations to Dr Cornelia Wiegand for her published clinical trial paper, “The efficacy of non-thermal gas plasma in the treatment of diabetic foot ulcers stalled by subclinical, biofilm-related wound infection”. This clinical trial paper features the use of our SteriPlas Cold Plasma medical device for the treatment of chronic DFUs infected with biofilm.
The parameters of this study were set much higher than a normal study, with there being a much higher standard of care given (3x per week) vs a typical outpatient setting of 1x weekly.
We wanted to show that even when chronic diabetic foot ulcers are subjected to more frequent outpatient visits with biopsies and swabs included (something not often collected during standard outpatient visits), SteriPlas Cold Plasma still prevails as the successful treatment method due to its physical mode of action and ability to kill all types of bacteria protected within biofilm.