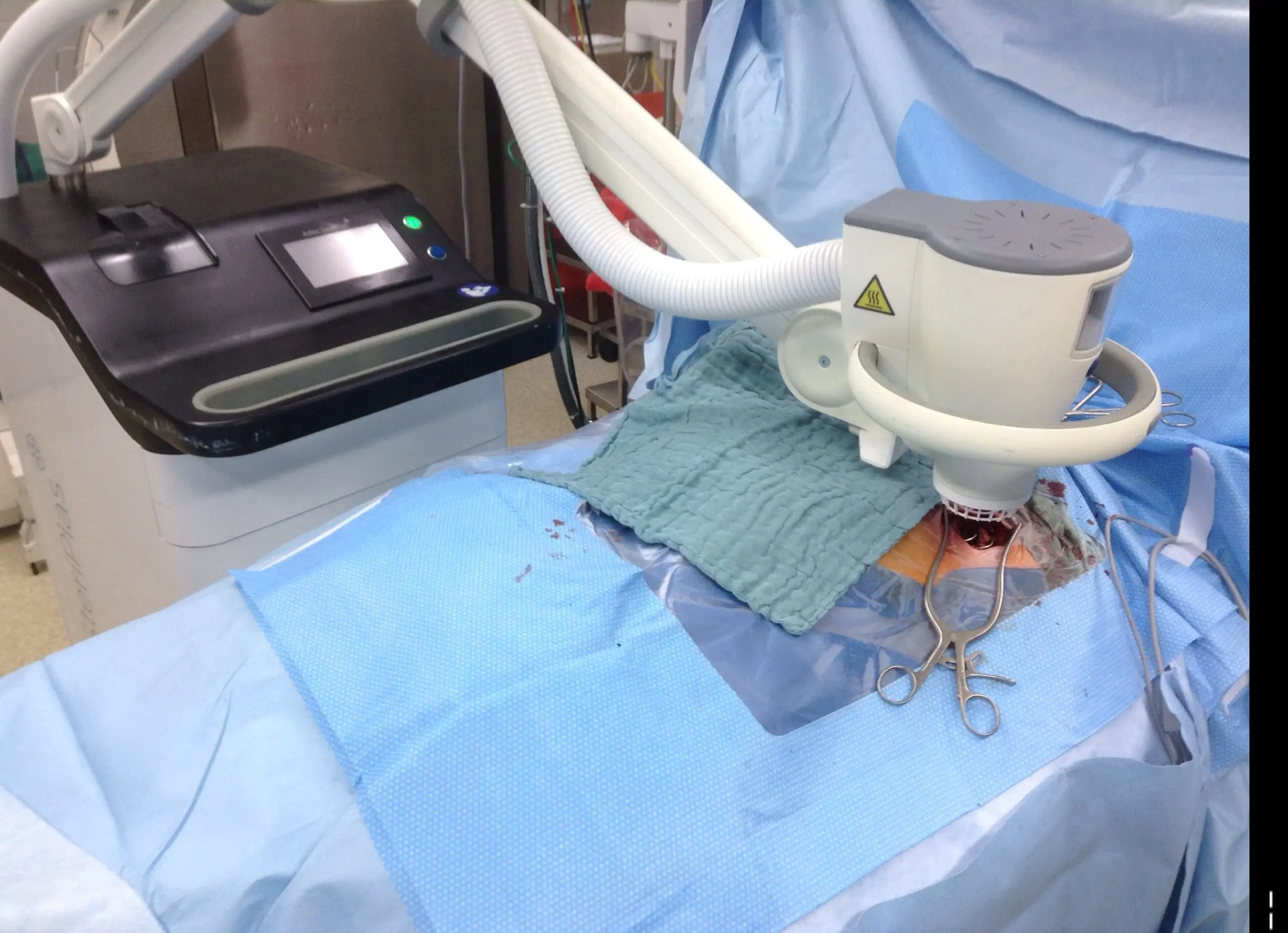
Deep Sternum Surgical Site Infections (DSSIs)
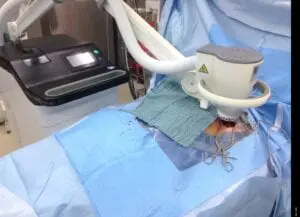
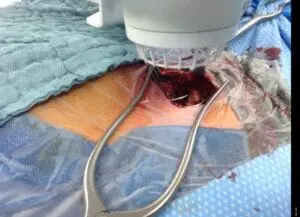
Deep sternal surgical site infections (DSSIs) are a still a severe complication after cardiac surgery. The mortality estimated range is between 15%-40% of all cases. In addition to the significantly extended hospitalization, there is a significant cost burden with a calculated cost of € 36,000 for a single DSSI case.
The use of the Adtec SteriPlas for these complicated conditions has been actively documented as better managing the chronic infections, reducing hospitalization times and cost but more importantly it has shown to decrease the mortality rate. Our medical device has been praised as a tissue and life saving medical device.
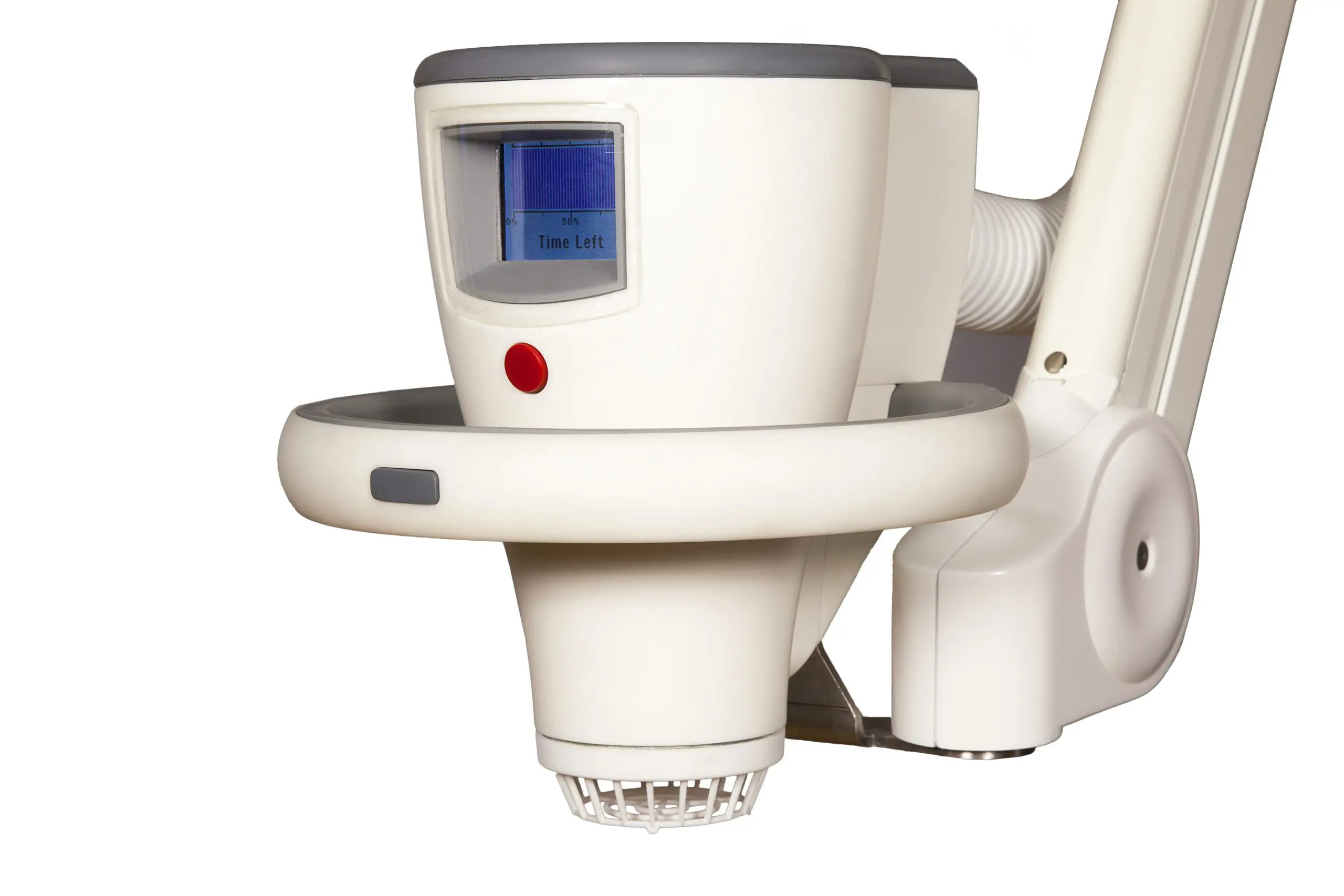
SteriPlas' patented 6 electrode plasma system
Did you know our plasma chamber features a patented 6 electrode plasma torch system? This is where the cold plasma is generated before it is propelled with the gas flow over the treatment site. Our 60+ peer reviewed clinical studies have shown that accelerated healing in stalled and non-healing wounds/skin conditions can be achieved with no side effects reported.
Herz medizin 2021

We look forward to supporting Dr Heinrich Rotering’s presentation, “Avoiding sternal wire removal in early onset of sternal site infection - Two years experience with an optimized treatment concept using cold atmospheric plasma“ at the Annual scientific meeting of the German Society for Thoracic, Cardiac and Vascular Surgery. His presentation will discuss the strong benefits of using the Adtec SteriPlas for complicated sternal wound infections in cardiac patients.
The conference will be held virtually between 26-28 February: https://www.dgthg.de/de/jahrestagung
SteriPlas cold plasma successful at killing multi-resistant bacteria
Our animation video shows how bacteria are easily destroyed using our patented cold plasma technology. The components of cold plasma work collectively to target bacteria and physically rupture the structure, allowing micropores to be created which gains access to the microbial DNA to be destroyed. This effectively kills bacteria, even if they are protected within biofilm.
Learn more about our clinical evidence by visiting our webpage www.adtechealthcare.com or send us an email at info@adtecplasma.com
Click the link below to access the animation video:
Animation video to show how the SteriPlas can successfully kill multi-resistant bacteria
Patient and user safety is priority with the Adtec SteriPlas
As a medical device company, we always strive to ensure the priority of patient and user safety. In comparison to battery-powered and smaller cold plasma devices which may produce unreliable and damaging levels of ozone and NOx, the Adtec SteriPlas has been carefully designed as a safe and well-balanced cold plasma medical device. It has been engineered to deliver predictable levels of cold plasma to ensure the continuity of no side effects reported, putting safety and efficacy at the top priority.
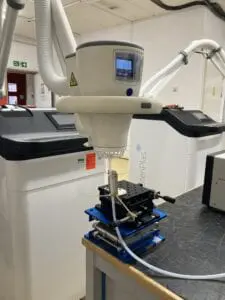

Adtec Healthcare celebrates yet another RCT publication

Adtec Healthcare celebrates another prospective, randomized and placebo-controlled trial (RCT) publication featuring our cold plasma medical device for the treatment of chronic wounds. The clinical trial conducted at the Essen University Medical Center in Germany and led by Professor Joachim Dissemond discusses the strong benefit of using the Adtec SteriPlas to treat chronic wounds. This is our first RCT measuring wound healing rates with respect to overall wound size using cold plasma.
The study consisting of three groups (Group 1 - plasma 1x/week, Group 2- plasma 3x/week and Group 3 - placebo 1x/week) shows the significant wound size reduction when using cold plasma vs placebo. Not surprisingly, Groups 1 and 2 led an astonishing 63.0% and 46.8% reduction in wound size for chronic wound patients, whereas Group 3 (placebo-treated) patients had increased in wound size by 17.5% larger.
The full publication can be viewed here: https://onlinelibrary.wiley.com/doi/epdf/10.1111/ddg.14294
The rich data of the RCT further demonstrates why our cold plasma medical device, the Adtec SteriPlas, continues to lead the way for cold plasma medicine by accelerating healing in non-healing and problematic wounds with the strong advantage of no side effects. The use of the Adtec SteriPlas is simple – a quick supplement to your standard treatment program for your patients.
Contact us at info@adtecplasma.com to learn more about our medical device and how to obtain this in your clinic.
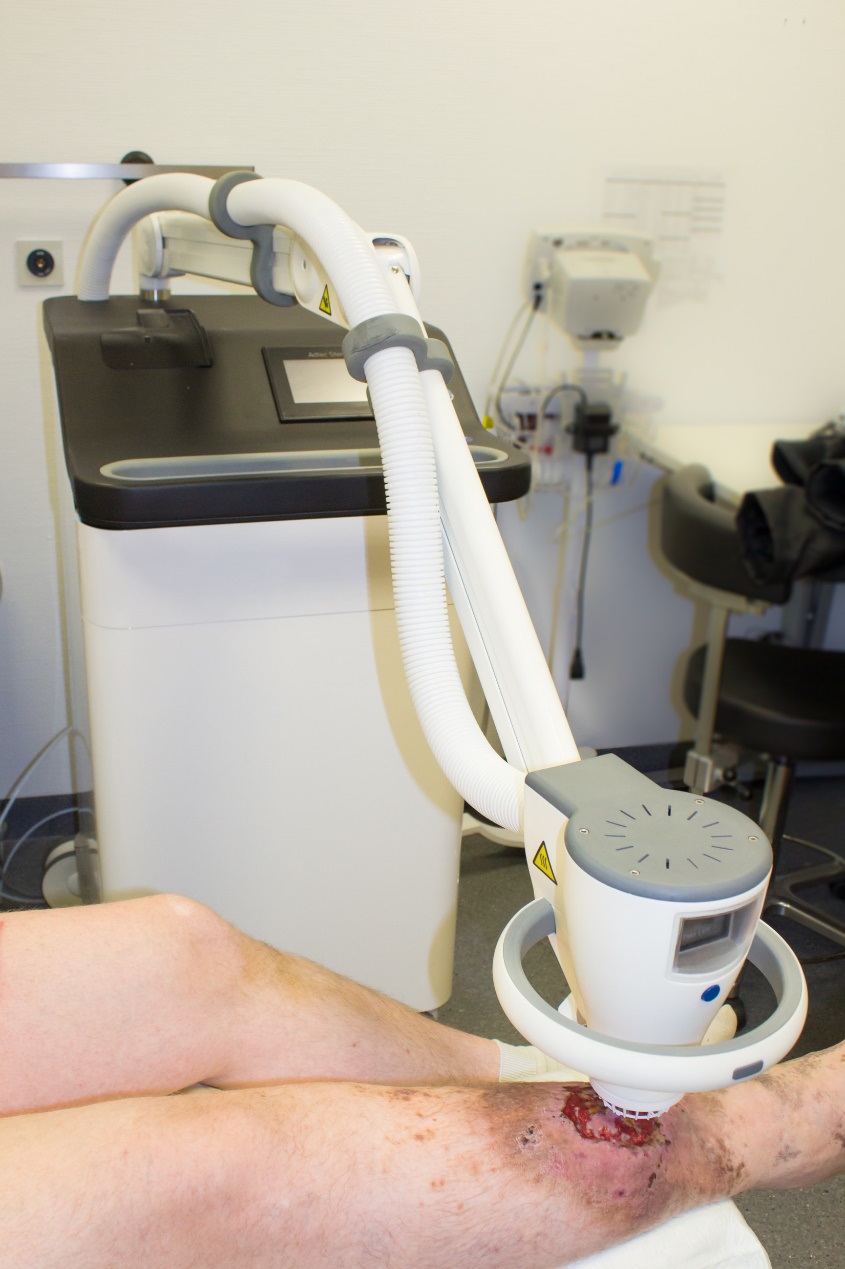
Leading cold plasma medical device
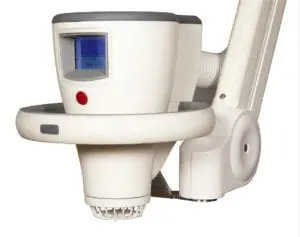
The Adtec SteriPlas is the leading cold plasma medical device used to manage the infection in chronic wounds, surgical site infections and to treat dermatological conditions such as acne and actinic keratoses.
With our wide clinical trials and publications bibliography, we are proud to state its strong antibacterial efficacy regardless of the type of bacteria or its resistance profile. There have been no side effects reported with the use of our medical device since it is safe, painless to touch and an effective method of treating patients from conventional therapies.
New Actinic Keratoses clinical trial publication release

Congratulations Professor Alexander Rösch and his team at the Department of Dermatology at the University Hospital Essen for their recent clinical trial publication. The study “Efficacy of cold atmospheric plasma versus diclofenac 3% gel in patients with actinic keratoses: a prospective, randomized, and rater-blinded study (ACTICAP)” can be found here.
Their clinical trial began in 2017 and its purpose was to test the efficacy of our medical device, the Adtec SteriPlas on actinic keratoses patients in comparison to those treated with Diclofenac 3% gel. The clinical efficacy of this modality is comparable to that of Diclofenac gel. However, unlike Diclofenac, cold atmospheric plasma showed no major side effects. Thus, this modality might be especially well-suited for patients who require nontoxic treatment options, particularly immunocompromised patients and those with extensive field cancerization. This continues to be one of our strongest and most favoured characteristics of the Adtec SteriPlas: no side effects.
We encourage you to read the clinical trial publication and welcome any questions that you have.
#actinickeratoses #actinickeratosis #skin #dermatology #coldplasma #kaltesplasma #clinicaltrial #sideeffects
Adtec Healthcare's new project with Hull York Medical School testing gas plasma on Osteomyelitis bone infections.
We are excited to announce the study “Gas Plasma for the Prevention and Management of Osteomyelitis Biofilms” has now begun at the Hull York Medical School. The study led by Dr Angela Oates and funded by the National Biofilms Innovation Centre will seek to test the efficacy of our gas plasma system on osteomyelitis bone infections using our PlasmaTact device. This project will develop a novel laboratory testing model to evaluate and optimise plasma treatment for osteomyelitis biofilm infections.

We have already demonstrated that our gas plasma medical device, the Adtec SteriPlas, has proven antibacterial efficacy and accelerated healing in problematic and non-healing wounds with strong evidence collected from our extensive library of clinical trials and publications. This includes diabetic foot ulcers which are often complicated by biofilm. Over the course of its existence it has shown that no side effects have been reported which offers patients a safe and reliable treatment option. It has also shown that a 2-minute treatment time is sufficient to achieve exceptional results and that it is also a broad spectrum antibacterial with the ability to kill a wide range of Gram-negative and Gram-positive bacterial superinfections.
The PlasmaTact utilizes the same gas plasma technology from the Adtec SteriPlas. The main difference between the two devices is that the PlasmaTact offers a smaller handheld style treatment area of 1cm2 (vs 12cm2 with the SteriPlas) and offers users the ability to change the power output settings (vs the fixed settings with the SteriPlas).
Osteomyelitis (OM) is biofilm infection of the bone and is a common and costly complication in diabetic foot ulcer (DFU) patients often resulting in amputations. Long-term antimicrobial therapy is widely used as a primary treatment for OM or as an adjunct to surgical approaches however, there is a failure rate of up to 35% and an associated increasing prevalence of antimicrobial resistance. There is an urgent clinical need to develop alternative non-antimicrobial approaches to treat infections, and currently there is a lack of credible alternatives in the market for the treatment of OM.
Evaluating the efficacy of gas plasma in OM represents unique challenges as both the biofilm growth and physical characteristics of OM bone will affect the delivery and activity of the plasma. In vitro biofilm model systems offer a means by which rapid, costs effective and standardised evaluation of anti-biofilm treatments can be undertaken.
The PATROL project is a new and exciting collaborative project between Adtec Healthcare and Dr Angela Oates of the University of Hull Wound Healing Group which is supported by NBIC PoC Award. The overall aim of this project is to develop an osteomyelitis biofilm infection model to support the optimisation and evaluation of a cold plasma technology for the management and prevention of OM.
This collaborative project represents the first phase of the novel utilisation of Adtec SteriPlas to treat OM or as an adjunct current therapy. Successful translation of this technology to treat OM will reduce antimicrobial usage and associated OM amputations, reducing NHS costs and ultimately improving patient outcomes.
#osteomyelitis #biofilm #boneinfection #infection #antimicrobial #antimicrobialresistance #amputation #plasma #gasplasma #coldplasma #kaltesplasma #NBIC #universityofhull #podiatry #diabeticfootulcer #diabeticfoot #diabetes #hullyorkmedicalschool
Adtec Healthcare confirms attendance for 3rd conference in 2020
Adtec Healthcare looks forward to exhibiting at the Diabetic Foot Study Group (DFSG) conference in Bratislava, Slovakia between 18-20 September 2020.
You can find us at booth 13 in the exhibition hall where our staff will be ready to welcome you. We will also have our flagship medical device, the Adtec SteriPlas, live on demonstration to show you how easy it is to treat diabetic foot ulcers.

#plasma #coldplasma #kaltesplasma #gasplasma #DFSG #DFSG2020 #diabeticfootulcer #wound #woundcare #woundhealing #wundkongress #wund #medicaldevice #DFSG #DFSG2020