Antimicrobial (antibiotic) Resistance – the drugs won’t work !
Antimicrobial resistance (AMR) poses a significant global threat of far-reaching proportions. It is estimated that drug resistant infections contribute to nearly 5 million deaths every year and predicted to increase to over 10 million deaths every year. WHO has declared that AMR is one of the top 10 global public health threats facing humanity.
(The worldwide total deaths from COVID is now just over 6 million and we all know how frightening it was before the vaccine was developed)
AMR occurs when bacteria, viruses, fungi and parasites change over time and no longer respond to medicines making infections harder to treat and increasing the risk of disease spread, severe illness and death. As a result, the medicines become ineffective and infections persist in the body, increasing the risk of spread to others. Microorganisms that develop antimicrobial resistance are sometimes referred to as “superbugs”. Without effective antimicrobials, the success of modern medicine in treating infections, including during major surgery and cancer chemotherapy, would be at increased risk. Chronic infections if left untreated could result in tissue damage, amputation, longer stays in hospitals, surgical interventions, or increased possibility of mortality. Patients who are infected with drug-resistant infections are more likely to develop complications and are up to three times more likely to die from the infection. Non-healing wounds in particular, are characterised by complex and mixed bacterial populations, often involving antibiotic-resistant bacteria as well as phenotypically tolerant bacteria in biofilm form. The biofilm factor is clearly of considerable clinical importance: it protects bacteria from antimicrobial agents leading to persistent and difficult to treat chronic infections, and it exacerbates the spread of antibiotic resistance. Surgical Site Infections are also linked to anti-microbial resistance.
SteriPlas cold plasma technology kills bacteria by a physical mode of action and bacteria are therefore unlikely to develop primary or secondary resistance, which we have documented from our clinical studies. SteriPlas cold plasma also kills antibiotic resistant bacteria (e.g. MRSA) and kills bacteria encased in biofilm which are typically up to 1000 times more resistant to antibiotics. SteriPlas has proven clinical efficacy in treating wound infections, diabetic foot infections and surgical site infections in all clinical studies, all with the bonus of no side effects reported.
SteriPlas cold plasma can be used to treat topical infections reserving antibiotics for severe systemic infections.
References
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(21)02724-0/fulltext
https://www.worldometers.info/coronavirus/
https://www.who.int/health-topics/antimicrobial-resistance
https://healthfirsteurope.eu/wp-content/uploads/2020/11/A3A4-48pp-Booklet-Spreads-1.pdf
Bowler, P., Murphy, C. & Wolcott, R. Biofilm exacerbates antibiotic resistance: Is this a current oversight in antimicrobial stewardship?. Antimicrob Resist Infect Control 9, 162 (2020).
Taiwan Wound Therapy Society Conference, March 2022




Sharing some photos from last weekend at the Taiwan Wound Therapy Society conference.
Our medical distributor, SG Biomedical, had a productive weekend demonstrating our SteriPlas to doctors and nurses keen to learn about the benefits of cold plasma for the treatment of wounds, surgical site infections and medical dermatology.
For more information about the Adtec SteriPlas and how to obtain this in the SE Asia region, please contact our distributor SG Biomedical for more information.
ToGCPiCPT

Congratulations to our Business Development Manager, Jeiram, for his published chapter in the “Textbook of Good Clinical Practice in Cold Plasma Therapy”. This prestigious book houses powerful material from esteemed authors in the cold plasma medicine field.
Jeiram’s chapter, “SteriPlas® and PlasmaTact®”, discusses the importance of Adtec Healthcare’s influence in the cold plasma medicine field from being the first company ever to conduct cold plasma clinical trials on wounds and to our modern day clinical efficacy in wounds, surgical site infections and medical dermatology skin conditions.
You can view this chapter here: https://doi.org/10.1007/978-3-030-87857-3_18
For more information about our cold plasma medical and non-medical devices, please contact us at info@adtecplasma.com
Our Cold Plasma Medical Device leads the way
Bacteria protected within biofilm are up to 1000x more resistant to antibiotics. This can complicate treatment options for wounds and surgical site infections but as it is an international form of therapy, it remains used throughout patient treatments despite it offering little to no help in healing problematic/stalled wounds and infections.
The cost burden associated to ongoing antibiotic therapy is significant as is the antimicrobial resistance rates that are ever growing. The World Health Organisation recognizes this and more hospitals are reaching out to alternative therapies to help curb the spending costs and antimicrobial resistance rates.
As the Adtec SteriPlas cold plasma has been shown to kill all forms of bacteria, regardless of their resistance profile or if they are Gram -ve or +ve or even if they are protected within biofilm, it has become widely adopted as an alternative to antibiotic therapy specifically in cases where patients are at a severe stage of infection such as those associated to diabetic foot ulcers and left ventricular assist device infections.
It has been praised as a life-saving medical device helping to heal infections that have been stalled for a very long time where conventional therapies have failed to act, reduce amputation and mortality rates.
This year we look forward to releasing a series of Health Economic publications documenting the cost effectiveness of our medical device against the use conventional therapies. We look forward to releasing this information to you in due time.
Until then, please reach out to us to learn more about why the interest in our medical device is growing daily. The data collection of our safety and efficacy shows why using an Adtec SteriPlas could completely change the way you treat patients.
Characterisation of a Cold Atmospheric Pressure Plasma Torch for Medical Applications
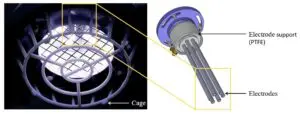
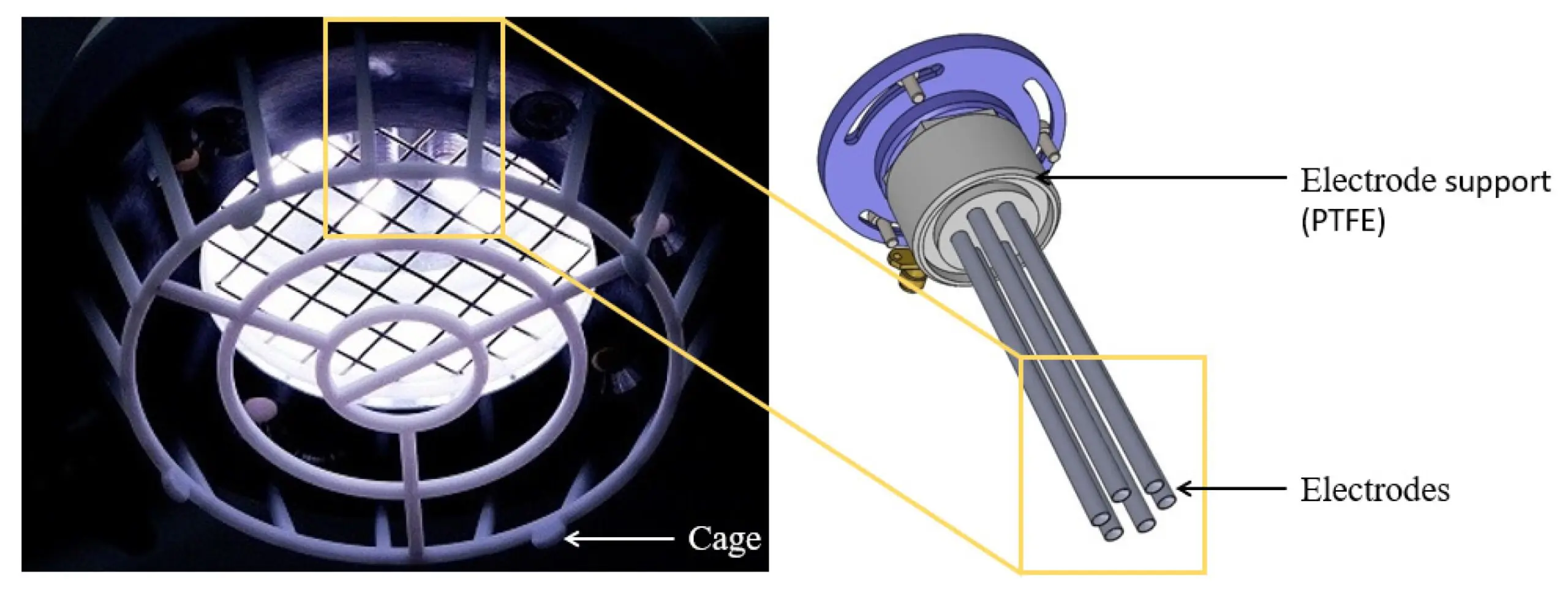
We congratulate Dr Adam Bennett on his most recent publication, “Characterisation of a Cold Atmospheric Pressure Plasma Torch for Medical Applications: Demonstration of Device Safety”.
The safety and effectiveness of plasma devices are of crucial importance, especially for applications where the plasma is discharged near humans. This study presents the novel design and characterisation of a Cold Atmospheric Plasma torch (SteriPlas), which is used in medical applications. The study shows the characterisation methodology that must be undertaken to show that a plasma device is safe, especially when used in an application on human skin. The emission spectrum discharged from the plasma torch is shown. The UV emitted is measured and the effective irradiance is calculated. The effective irradiance enables the determination of the maximum UV exposure limits, which in this application are shown to be over two hours; however, in some applications may be only seconds. NOx and ozone emissions are also recorded. The NOx levels in this application are shown to be orders of magnitude lower than their safety limits and the ozone emissions are also shown to be safe; however, in some plasma technologies the NOx and ozone levels are orders of magnitude higher than the safe levels.
This paper concludes with a discussion of how safety limits vary in different regions around the world and proposes an international standard. It documents the safety of our medical device which further reiterates one of our main strengths where no side effects have been reported.
Access to the full paper can be found here: https://www.mdpi.com/2076-3417/11/24/11864
E-poster at the EADV 2021 Conference

We look forward to supporting Fiona Koch’s e-poster that will be live during the EADV 2021 Spring Symposium tomorrow. Tune in to their website to get full access to her poster, “Efficacy of cold atmospheric plasma versus diclophenac 3% gel in patients with actinic keratoses: a prospective, randomized and rater-blinded study (ACTICAP)”.
To learn more about the Adtec SteriPlas’ strong clinical efficacy for the treatment of actinic keratoses, contact us at info@adtecplasma.com
The SteriPlas sets foot in the U.S.A.

We are excited to share news of our collaboration with The Southern Illinois University in United States of America. Our Adtec SteriPlas will be used as part of their study to examine the effect of cold plasma on a variety of infection burn model cases in a research laboratory setting. This study aims to help tackle the outbreaks of antimicrobial resistant microorganisms that continues to be an issue for burns cases. We look forward to providing more information and the results as the study progresses.
Plastische Chirurgie features the Adtec SteriPlas
Be sure to read our latest advertorial in the Plastische Chirurgie magazine featuring the Adtec SteriPlas for the treatment of actinic keratoses. Our medical device has been thoroughly tested in clinical trials and studies to prove its efficacy for the treatment of complex and impaired wounds, surgical site infections and medical dermatology conditions leading to accelerated healing.
Existing conventional therapies currently on the market for actinic keratoses have sometimes been shown to bear undesirable side effects. So we are proud to boast the safety and reliability of the Adtec SteriPlas with no side effects reported over continuing use since its existence especially for the treatment of actinic keratoses lesions.
To learn more about our medical device, send us an email at info@adtecplasma.com

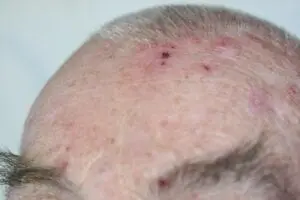
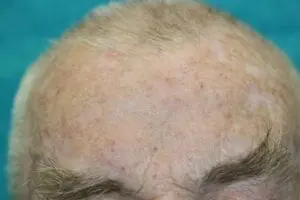
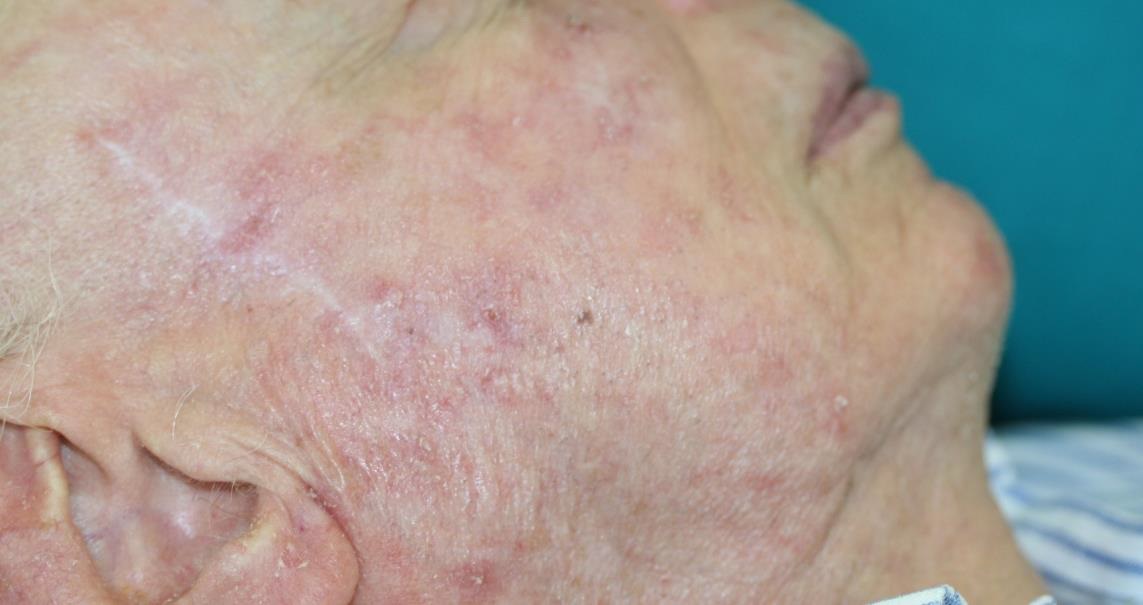
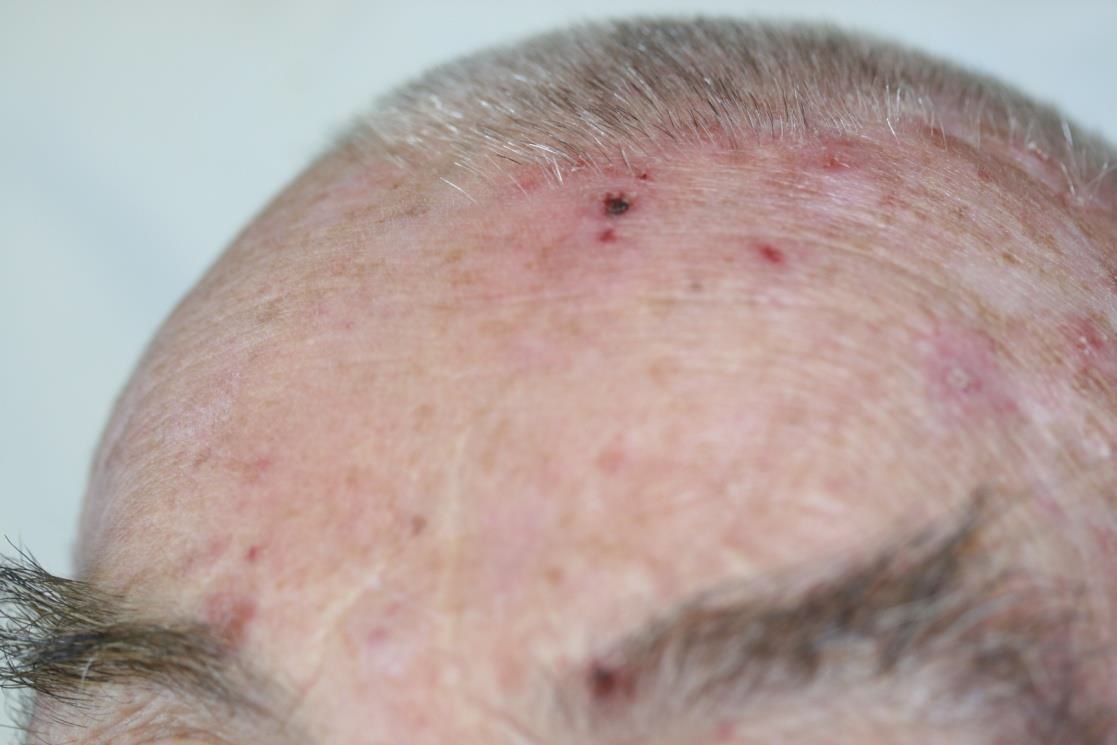
The SteriPlas can be used to treat a variety of areas on the body
We are proud have been able to treat a wide scale of body areas. Our clinical evidence ranges across a multitude of conditions and areas of the body including the foot, leg, arms, head, neck, scalp, hands, axilla and even groin. All wounds/lesions present in these areas of the body had shown positive responses to treatment with the Adtec SteriPlas.
We are always open to treating new conditions and welcome any study proposals that you have. If you would like to get the Adtec SteriPlas in your clinic or hospital, please contact us at info@adtecplasma.com where one of our team members will assist you with the delivery.
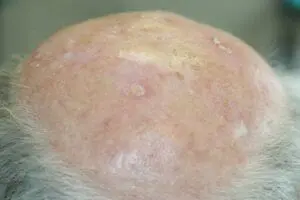
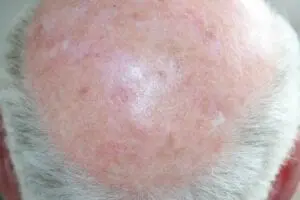
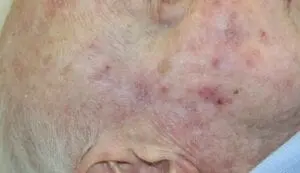
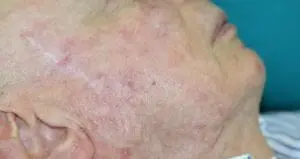
The Adtec SteriPlas featured in another Actinic Keratoses publication
We would like to congratulate Dr Mariachiara Arisi and her team at The Department of Dermatology, Brescia Hospital in Italy for their latest publication: “Cold Atmospheric Plasma (CAP) for the Treatment of Actinic Keratosis and Skin Field Cancerization: Clinical and High-Frequency Ultrasound Evaluation”.
This publication features the Adtec SteriPlas and further demonstrates its efficacy for treating complicated Actinic Keratoses (AK) lesions. All patient enrolled into this study had previously shown resistance or intolerance to conventional field-directed therapies. The results of the study show how all patients who received treatment with the Adtec SteriPlas had significantly reduced AK lesions with the benefit of no side effects observed, quite the opposite for conventional therapies that often bear undesirable side effects. The Adtec SteriPlas continues to be a favoured treatment alternative for actinic keratoses by continuing to be faster-acting, safer and reliable.
You can read the full publication here: https://link.springer.com/article/10.1007%2Fs13555-021-00514-y