Taiwan Wound Therapy Society Conference, March 2022




Sharing some photos from last weekend at the Taiwan Wound Therapy Society conference.
Our medical distributor, SG Biomedical, had a productive weekend demonstrating our SteriPlas to doctors and nurses keen to learn about the benefits of cold plasma for the treatment of wounds, surgical site infections and medical dermatology.
For more information about the Adtec SteriPlas and how to obtain this in the SE Asia region, please contact our distributor SG Biomedical for more information.
Our patented 6 electrode Cold Plasma Torch

The Adtec SteriPlas features a patented 6 electrode cold plasma torch. It’s proven efficacy for the destruction of all forms of bacteria including multi-resistant bugs makes it an alternative to antibiotic therapy for the treatment of wounds, surgical site infections and medical dermatology skin conditions. This is due to the unique physical mode of action delivered with our cold plasma as opposed to the chemical mode of action demonstrated with antibiotics. Broader studies have shown that antimicrobial resistance is unlikely to be generated with cold plasma treatment because of this.
A huge collection of our peer reviewed publications documents the clinical efficacy and safety of our medical device across a broad range of conditions including diabetic foot ulcers, left ventricular assist device (LVAD) infections, deep sternal wound infections, actinic keratoses and acne.
To learn more about our medical device or to obtain one in your clinic practice, please contact us at info@adtecplasma.com
Characterisation of a Cold Atmospheric Pressure Plasma Torch for Medical Applications

We congratulate Dr Adam Bennett on his most recent publication, “Characterisation of a Cold Atmospheric Pressure Plasma Torch for Medical Applications: Demonstration of Device Safety”.
The safety and effectiveness of plasma devices are of crucial importance, especially for applications where the plasma is discharged near humans. This study presents the novel design and characterisation of a Cold Atmospheric Plasma torch (SteriPlas), which is used in medical applications. The study shows the characterisation methodology that must be undertaken to show that a plasma device is safe, especially when used in an application on human skin. The emission spectrum discharged from the plasma torch is shown. The UV emitted is measured and the effective irradiance is calculated. The effective irradiance enables the determination of the maximum UV exposure limits, which in this application are shown to be over two hours; however, in some applications may be only seconds. NOx and ozone emissions are also recorded. The NOx levels in this application are shown to be orders of magnitude lower than their safety limits and the ozone emissions are also shown to be safe; however, in some plasma technologies the NOx and ozone levels are orders of magnitude higher than the safe levels.
This paper concludes with a discussion of how safety limits vary in different regions around the world and proposes an international standard. It documents the safety of our medical device which further reiterates one of our main strengths where no side effects have been reported.
Access to the full paper can be found here: https://www.mdpi.com/2076-3417/11/24/11864
Successful meetings held at the Taiwan Society for Plastic Surgery






We are happy to report an eventful exhibition over the weekend at the Taiwan Society for Plastic Surgery led by our distributor in Taiwan. This is the first time the Adtec SteriPlas has set foot in Taiwan so naturally there was a lot of interest in our medical device. We look forward supporting our distributor for the exciting year ahead.
For more information about our medical device, contact us at info@adtecplasma.com
The first cold plasma medical device to treat Scleroderma

We congratulate Dr Stephanie Ardnt and Prof. Sigrid Karrer from the University Hospital Regensburg for their recent publication, “The Anti-Fibrotic Effect of Cold Atmospheric Plasma on Localized Scleroderma In Vitro and In Vivo”.
This publication features the use of our Adtec SteriPlas and shows strong efficacy for the treatment of Scleroderma, which up to now has not yet been evaluated by CAP. The extensive study documents significantly reduced dermal thickness and collagen deposition as well as a decrease in both alpha smooth muscle actin-positive myofibroblasts and CD68-positive macrophages in the affected skin in comparison to untreated fibrotic tissue. This study provides the first evidence for the successful use of CAP for treating LS and may be the basis for clinical trials including patients with LS.
https://doi.org/10.3390/biomedicines9111545
Deep Sternal Wound Infections (DWSI) and the Adtec SteriPlas

Unlike other cold plasma medical devices currently present on the market, the Adtec SteriPlas is not limited to the treatment of superficial wounds only.
For example, deep sternal wound infections (DSWIs) are a potentially devastating complication of median sternotomy performed in cardiac surgery. These large wound cavities are prone to infections and there is a concerningly high morbidity and mortality rate associated to these patients. The use of conventional therapies alone has shown to not always be successful and expensive muscle flap surgery is usually required.
The large 12cm2 treatment area of the Adtec SteriPlas covers a greater surface area of the wound compared to its competitors. This allows the benefit of a quick 2 minute treatment time for every 12cm2 covered. Because of its propulsion towards the treatment site, the cold plasma components work quickly to defeat bacteria and allow the management of the infection. The use of the Adtec SteriPlas has shown to accelerate healing rates and prevent the need for expensive surgery options that would otherwise have been needed.
It has been praised as a lifesaving medical device and is now widely used across multiple hospitals for the management of DWSIs.
Safe UV and Reactive Species levels only with the Adtec SteriPlas
As a leading medical device company, the crucial balance of safety and clinical efficacy are a top priority. We are always conducting tests to demonstrate the approved safety and reliability of the Adtec SteriPlas but to also learn new ways to improve the use of cold plasma in medicine.
Our clinical studies already validate the safety of the Adtec SteriPlas, including its low-level UV and reactive species produced from the cold plasma. These two components are imperative for the physical destruction of bacteria, however, too much of a good thing can also be relatively damaging like that observed with cold plasma jet/pens and battery powered cold plasma devices.
Measured across multiple distances, the UV light produced is well below the ICNIRP limit of 30 J/m2 and the reactive species produced are well below the occupational limits set by NIOSH and UK HSE. The Adtec SteriPlas continues to be one of the only cold plasma medical devices that puts patient and user safety first with no reported side effects and promising clinical efficacy from every treatment conducted.

For more information about the Adtec SteriPlas, send us an email at info@adtecplasma.com
Casopis Lecba ran-page-003
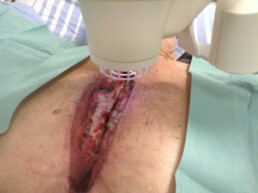
Did you see the Adtec SteriPlas featured in the Czech Society for Wound Healing magazine?
Dr Hokynková from the Department of Burns and Plastic Surgery at Brno Hospital in Czech Republic had published a case study in the magazine to document the efficacy of the Adtec SteriPlas after having tested this on a variety of wounds including pressure ulcers and diabetic foot ulcers.
The article validates the SteriPlas’ ability to significantly reduce microbial load within the wound which allows the body to regain control and accelerate healing of stalled wounds. This includes wounds that are already contaminated with biofilm and listed for amputations. A change from the conventional therapies already existing on the market, the Adtec SteriPlas has proven to reverse further deterioration of complicated wounds and allow full healing and stability to be achieved.
For a translation of the article or to learn more about our efficacy and treatment options, please email us at info@adtecplasma.com



The SteriPlas sets foot in the U.S.A.

We are excited to share news of our collaboration with The Southern Illinois University in United States of America. Our Adtec SteriPlas will be used as part of their study to examine the effect of cold plasma on a variety of infection burn model cases in a research laboratory setting. This study aims to help tackle the outbreaks of antimicrobial resistant microorganisms that continues to be an issue for burns cases. We look forward to providing more information and the results as the study progresses.
Plastische Chirurgie features the Adtec SteriPlas
Be sure to read our latest advertorial in the Plastische Chirurgie magazine featuring the Adtec SteriPlas for the treatment of actinic keratoses. Our medical device has been thoroughly tested in clinical trials and studies to prove its efficacy for the treatment of complex and impaired wounds, surgical site infections and medical dermatology conditions leading to accelerated healing.
Existing conventional therapies currently on the market for actinic keratoses have sometimes been shown to bear undesirable side effects. So we are proud to boast the safety and reliability of the Adtec SteriPlas with no side effects reported over continuing use since its existence especially for the treatment of actinic keratoses lesions.
To learn more about our medical device, send us an email at info@adtecplasma.com